Migration studies
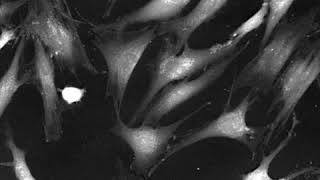
The mechanisms of cell migration largely depend on cell type and the guiding cellular environment where cells adapt their microenvironment to migrate efficiently. Innovative microscopic technologies allowing for live-cell imaging open a new horizon for studying cell migration in 2D and 3D environments.
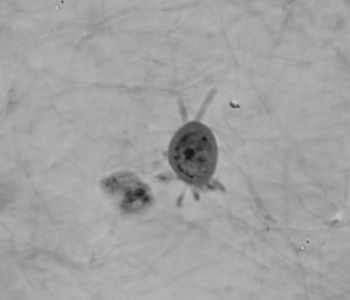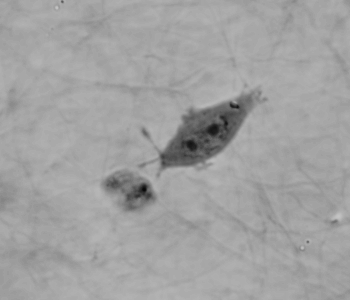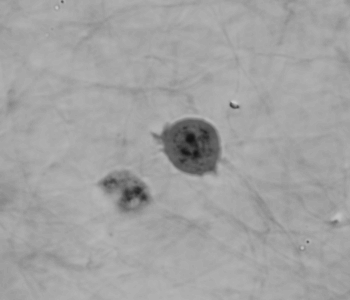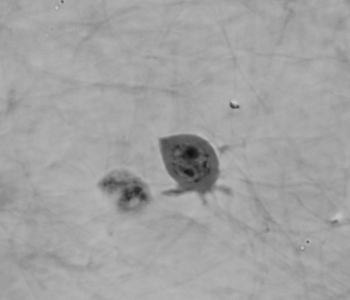
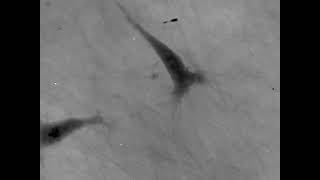
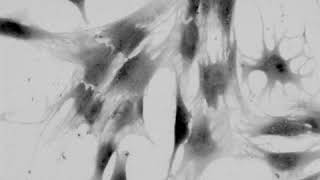
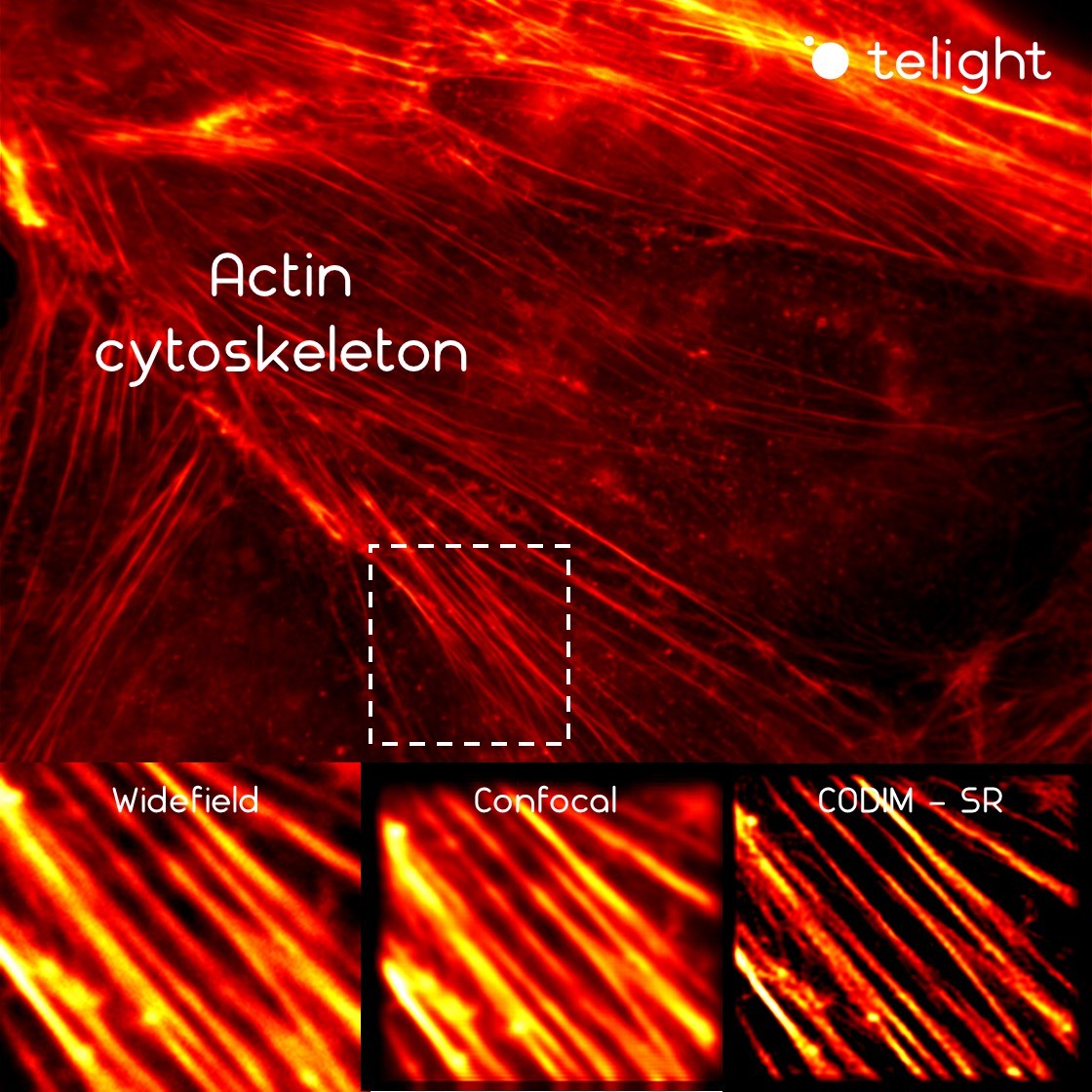
Cell migration mechanisms largely depend on cell type and the guiding cellular environment. Thus, cells acquire specific migratory phenotypes to migrate efficiently through tissues and adapt to their microenvironment. Motile cells are polarized, and the actin cytoskeleton is a key modulator of cell polarity and pseudopods whose stabilization determines the directionality of the movement.
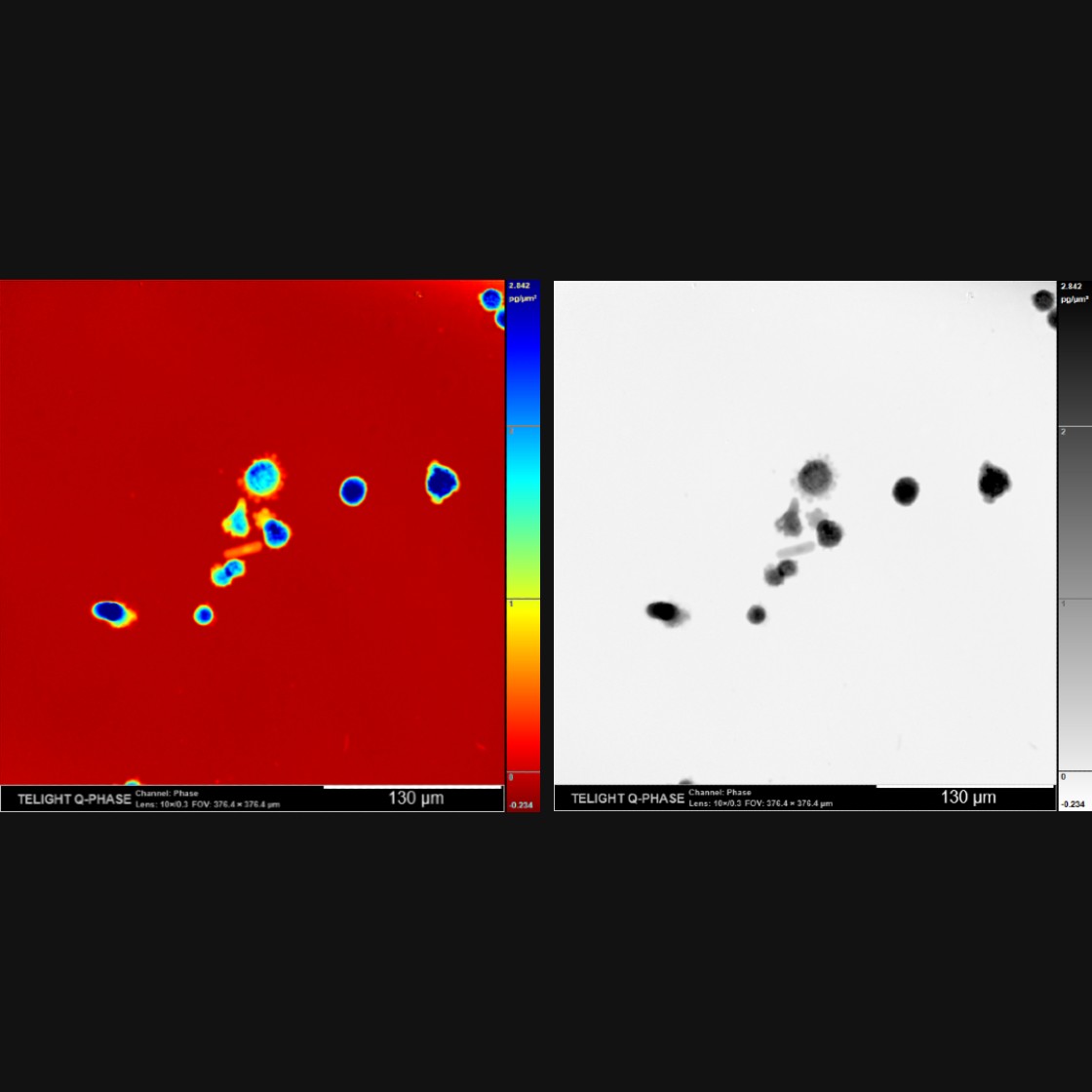
Q-Phase defines the pattern of cell polarity through the assessment of the morphological profile of migratory cells together with the measurement of their core and peripheral dry mass distribution.
Deregulation of cell migration is involved in cancer progression and metastatic development. Thus it constitutes a major research topic of interest for decades already.
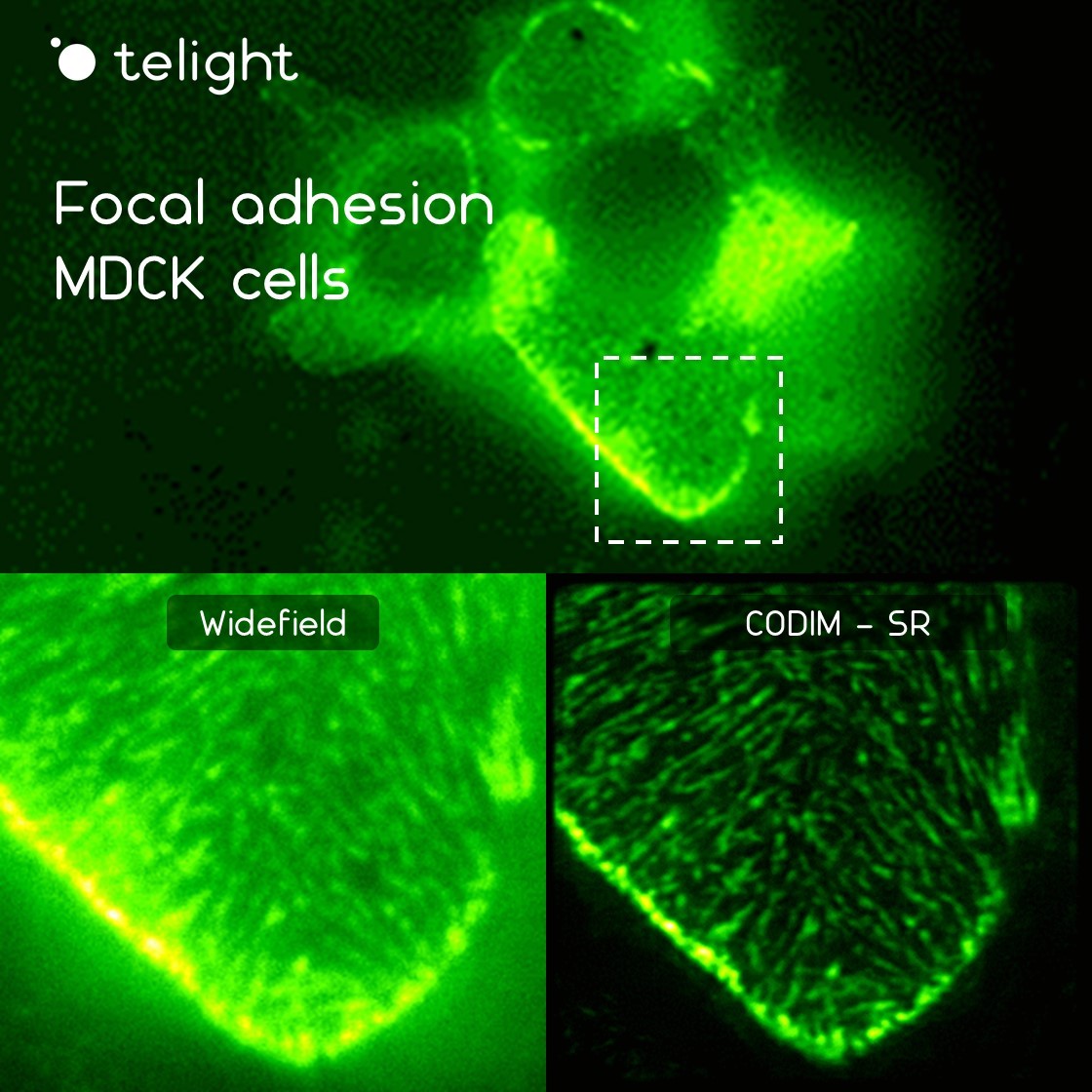
In this context, the LiveCodim can represent the perfect choice to dynamically image the actin cytoskeleton and visualize the specific structures involved in cell migration and invasion in superresolution, such as actin stress fibers, filopodia, lamellipodia, endosomes, etc.
Publications
Quantitative phase imaging unravels new insight into dynamics of mesenchymal and amoeboid cancer cell invasion
M. Fojtů, et al.
Silicane Derivative Increases Doxorubicin Efficacy in an Ovarian Carcinoma Mouse Model: Fighting Drug Resistance
Cisplatin enhances cell stiffness and decreases invasiveness rate in prostate cancer cells by actin accumulation
O. Ramaniuk, et al.
Quantitative Phase Imaging of Spreading Fibroblasts Identifies the Role of Focal Adhesion Kinase in the Stabilization of the Cell Rear
Products

Q-Phase
Quantitative Phase Imaging
Q-Phase is a patented holographic microscope with high detection sensitivity, designed for gentle live-cell imaging.
Q-Phase is an ideal solution for experts who desire precise automated segmentation of individual cells for subsequent data analysis. Q-Phase quickly transforms cell features and dynamics into numerical data suitable for comparisons, correlations, and more detailed statistics.

LiveCodim
From conventional to super-resolution microscopy
LiveCodim is a universal, super-resolution imaging platform designed to interface with any standard fluorescence microscope. It is the solution for live-cell imaging with high resolution and low phototoxicity.