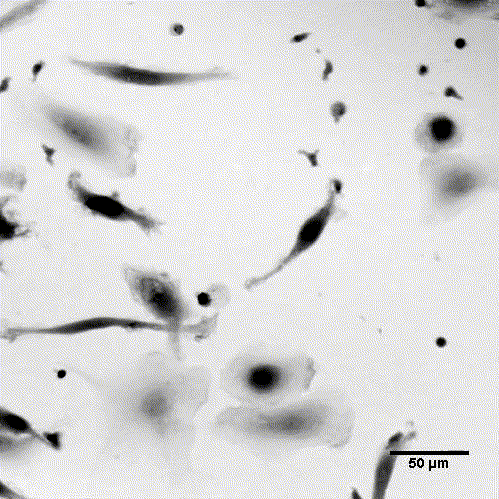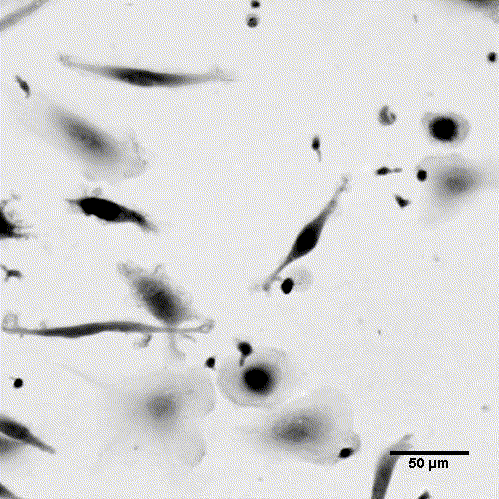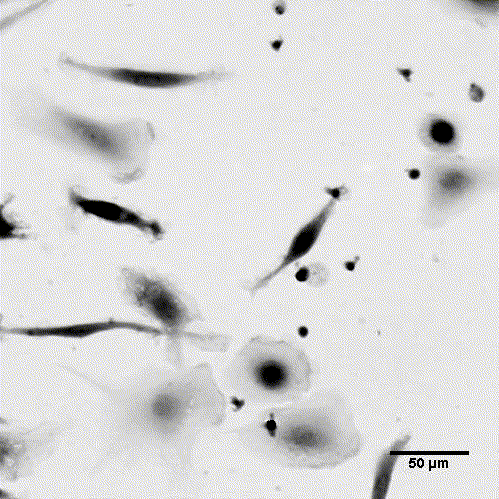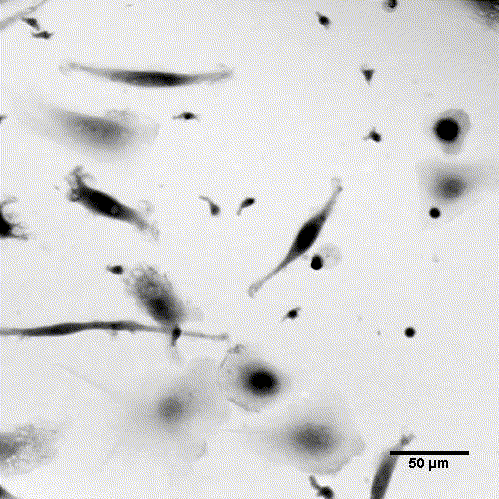
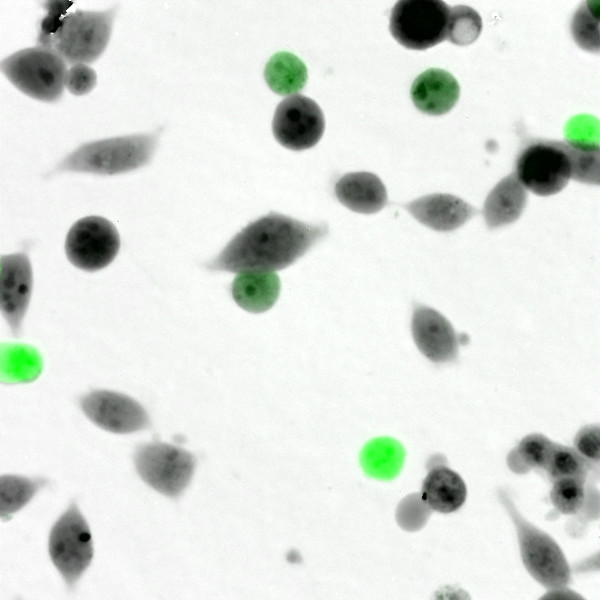
LiveCodim
From conventional to super-resolution microscopy
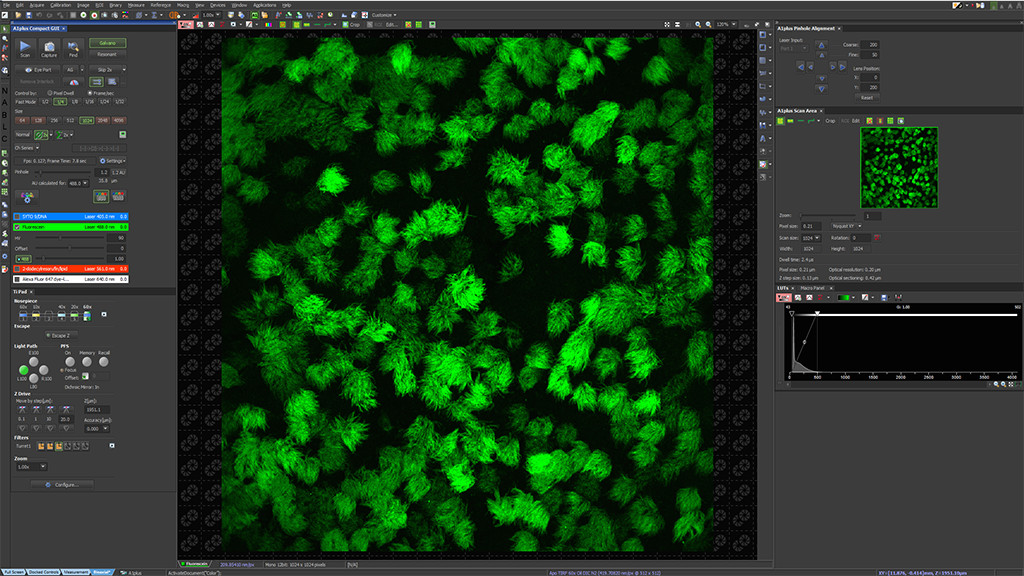
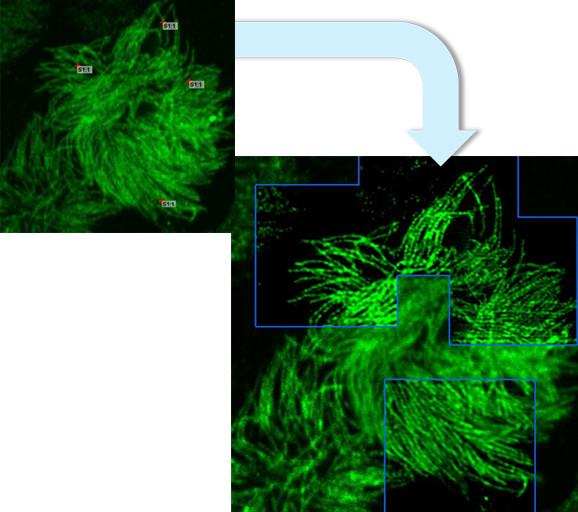
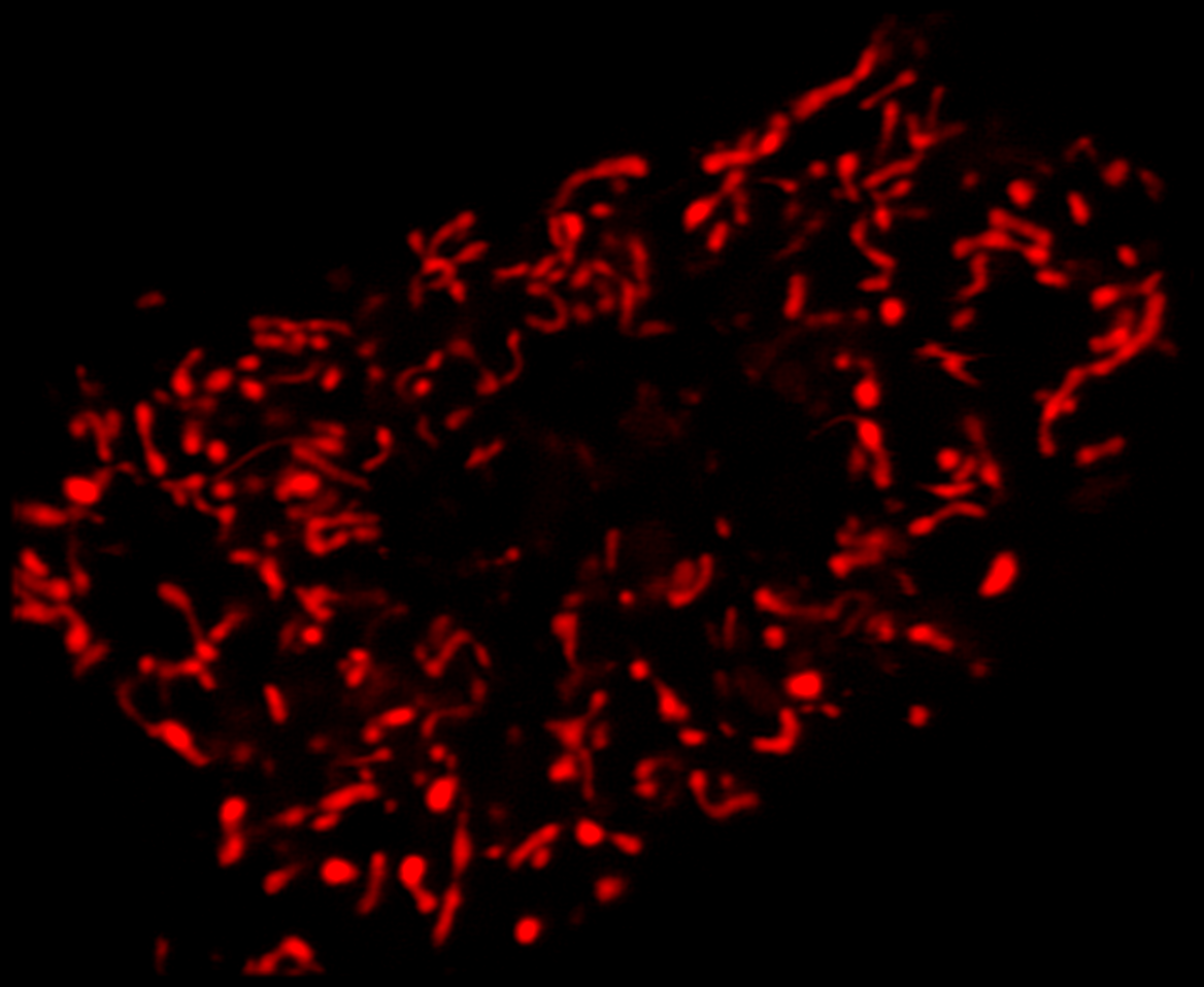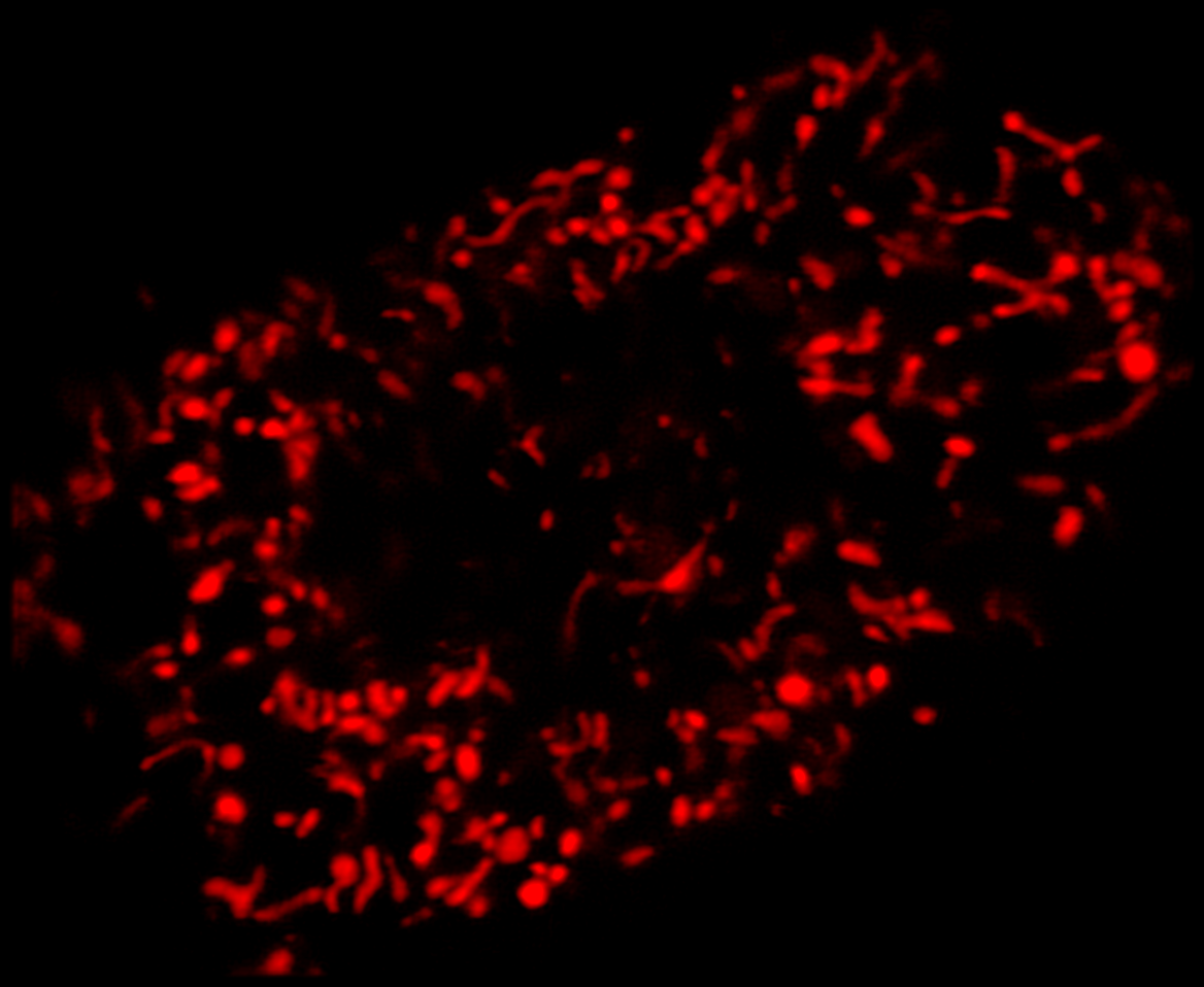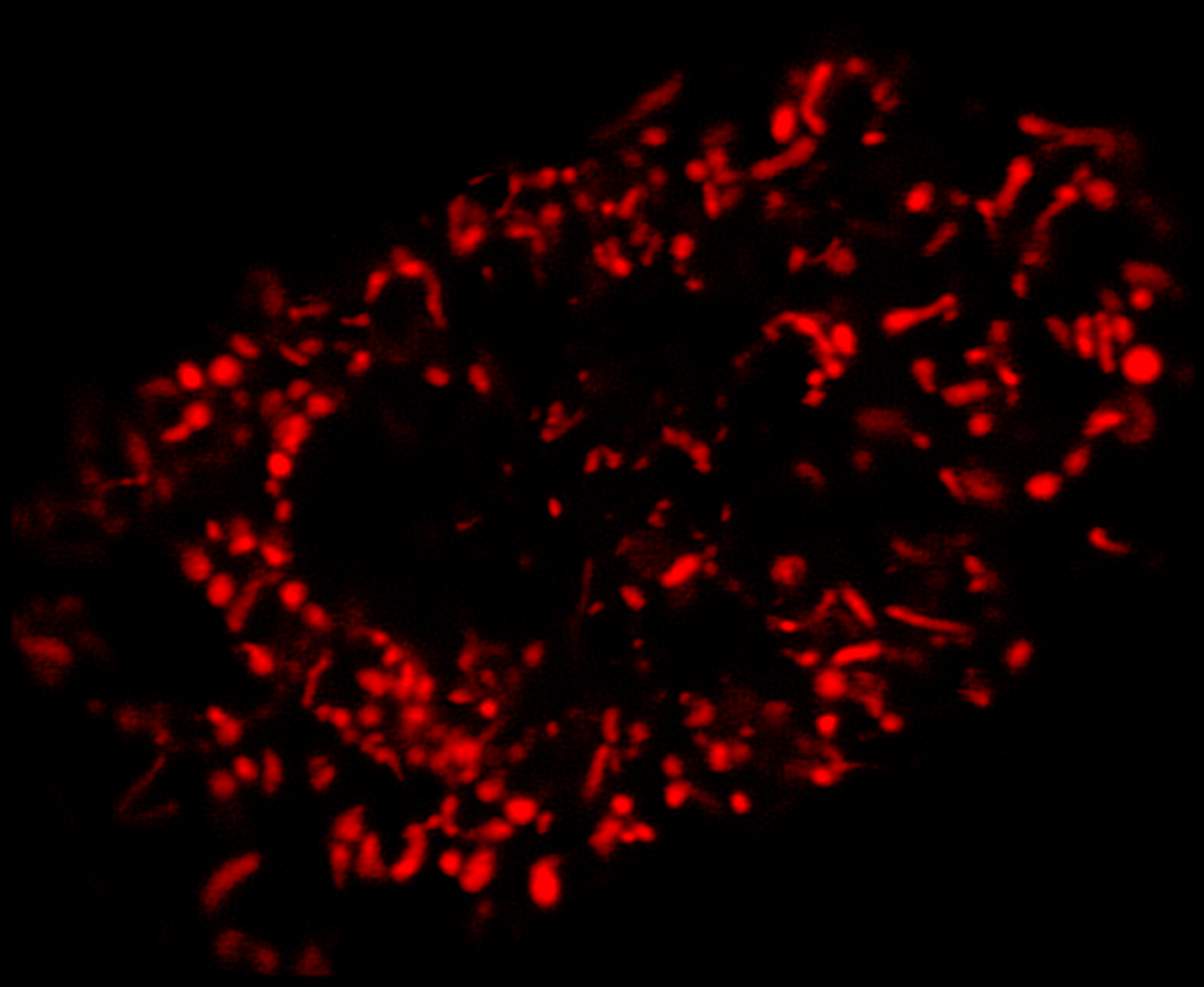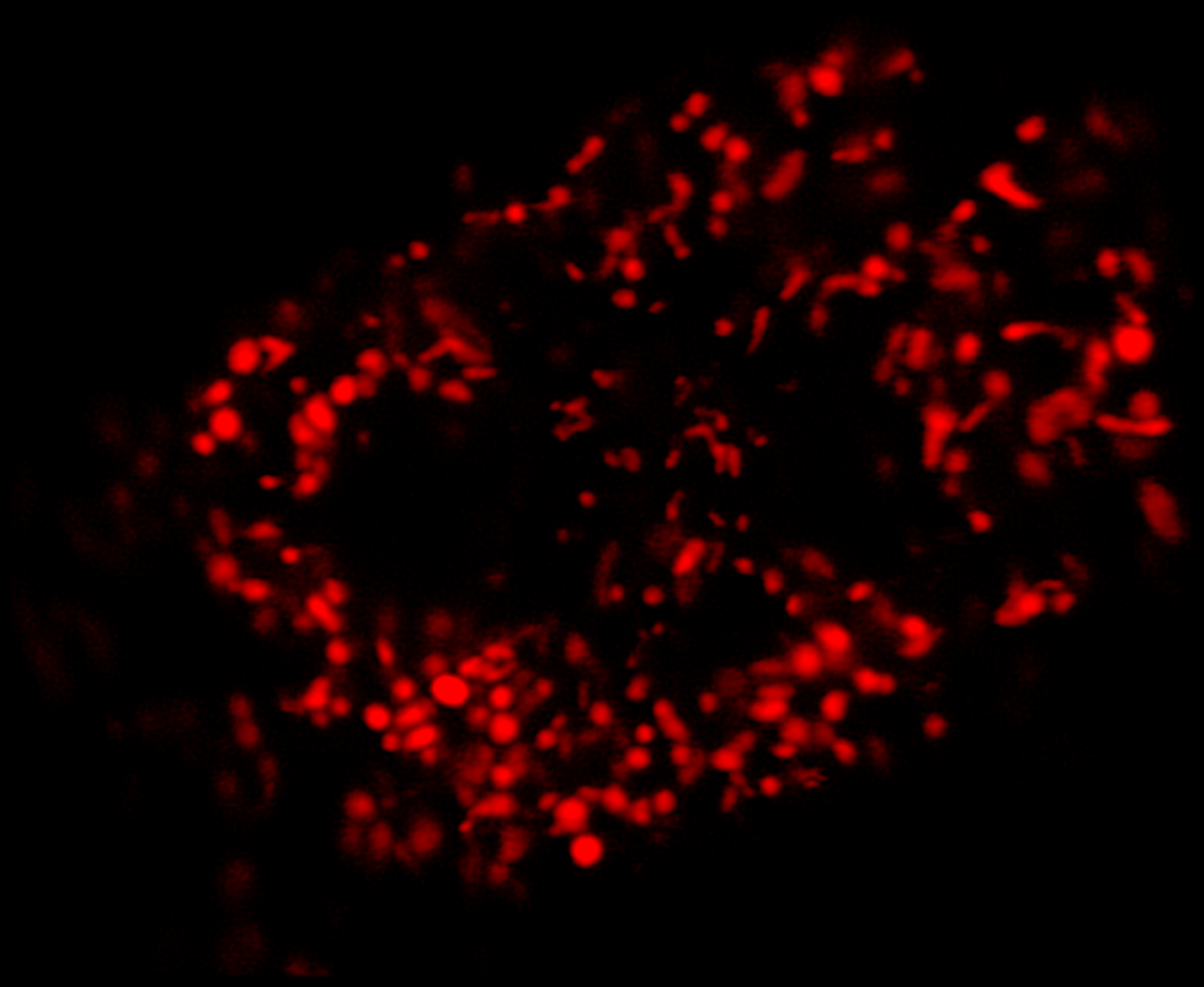
LiveCodim is a universal, super-resolution imaging platform designed to interface with any standard fluorescence microscope. It is the solution for live-cell imaging with high resolution and low phototoxicity.

Description
LiveCodim establishes a new super-resolution imaging modality, creating an answer for the main critical challenges of SR in cell biology: maintaining the integrity of biological samples, technical complexity, cost of ownership.
LiveCodim consists of a universal add-on, for any standard microscope. It includes widefield, confocal, and super-resolution modalities to provide a complete solution, especially suitable for retrofitting.
The technology is based on the patented Conical Diffraction Microscopy (CODIM) - a powerful beam shaper, generating controlled and localized light patterns used to scan biological samples with very low phototoxicity and negligible photobleaching.

Workflow
Set your regular live observation on the confocal microscope and optimize your image acquisition settings.
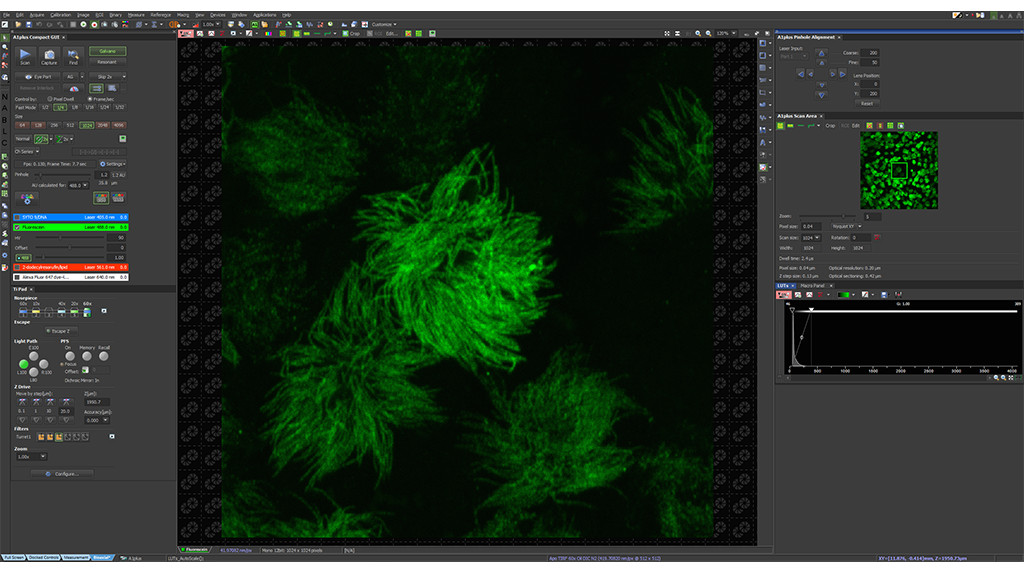
Position the desired area at the center of the full field of view and perform a 5x confocal zoom-in.
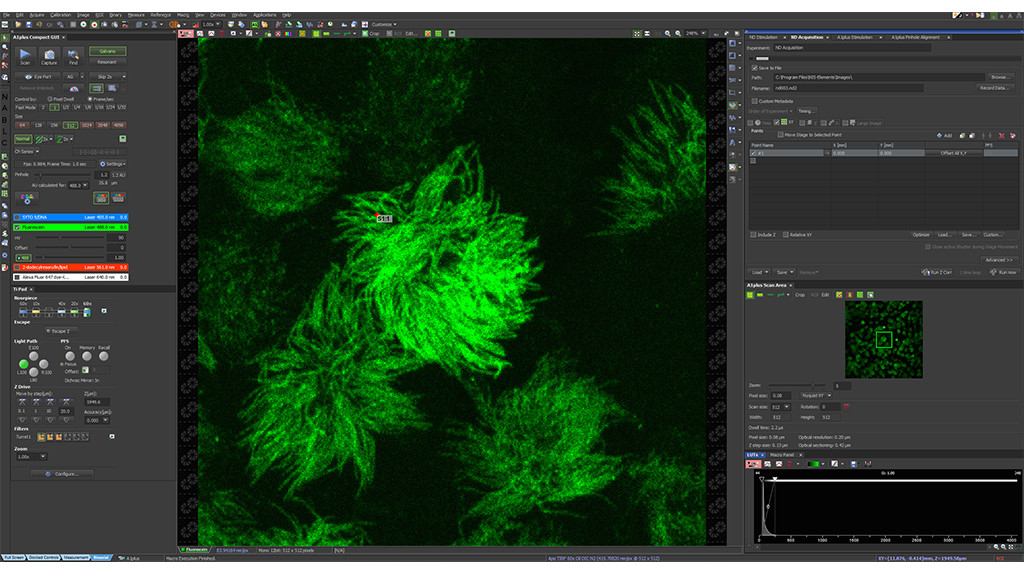
Select your region of interest to image in super-resolution mode and define CODIM acquisition settings.
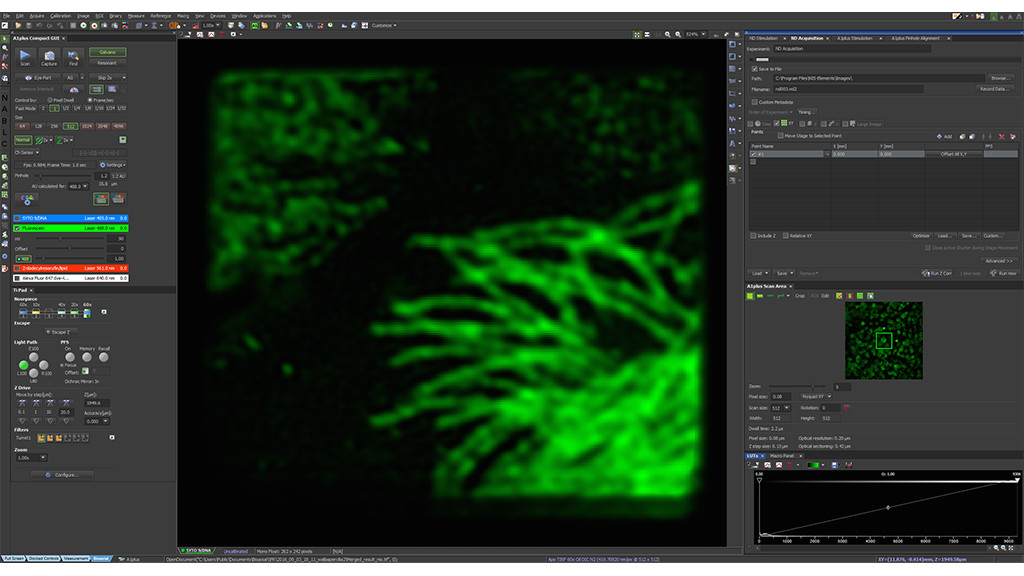
Start CODIM super-resolution acquisition then acknowledge the result in the confocal window.
Repeat the process over and over to inspect multiple areas.
Key advantages
RESOLUTION better or equal 90 nm XY | Increased resolution using light structuration and scanning point approach
High-performance SUPER-RESOLUTION MULTICOLOR IMAGING | 4 fluorescence channels
LIVE-CELL IMAGING ׀ Low phototoxicity and low photobleaching enabling in vivo SR study
PENETRATION DEPTH ׀ Z-stack ability and "suppression" of signal from other planes
STANDARD SAMPLE PREPARATION ׀ Standard fluorescence microscopy sample preparation workflow
SEAMLESS INTEGRATION ׀ with all fluorescence microscopes
Streamlined and EASY-TO-USE application ׀ Autonomous processing algorithm with no need for user-adjustment of many parameters
Download brochure
Publications
J. Vargas, et al.
The Wnt/Ca2+ pathway is involved in interneuronal communication mediated by tunneling nanotubes
Maarifi, G., Fernandez, J., Portilho, D.M., et al.
RanBP2 regulates the anti-retroviral activity of TRIM5α by SUMOylation at a predicted phosphorylated SUMOylation motif
Garita-Hernandez M., et al.
Optogenetic light sensors in retinal organoids
Getz A.M., Xu F., Visser F., et al.
Tumor suppressor menin is required for subunit-specific nAChR Alpha5 transcription and nAChR-dependent presynaptic facilitation in cultured mouse hippocampal neurons
Technology

“We traded all the beauty and elegance of Poggendorff rings and conical diffraction for a dull but efficient controllable beam shaping unit."
(Gabriel Y. Sirat)
Conical diffraction beam shaper
In 2006, Gabriel Y. Sirat demonstrated that conical diffraction can be used as a practical tool to shape optical beams, and suggested creating a versatile beam shaper and apply it to super-resolution fluorescence microscopy.
A thin biaxial crystal transforms the Point Spread Function (PSF) of a regular incident beam into an extensive family of light distributions, the choice of the distribution being controlled by the input and output polarizations.
Since 2019 BioAxial goes on with development as a part of Telight group.
Practically, beam shaping is achieved by enclosing the biaxial crystal between controllable polarizers; this simple optical set-up, similar to a polarimeter, has the ability to switch from one pattern to another pattern with a different topology in microseconds – or even faster. In addition, these patterns are perfectly co-localized, as they produced by the same primary optical beam.
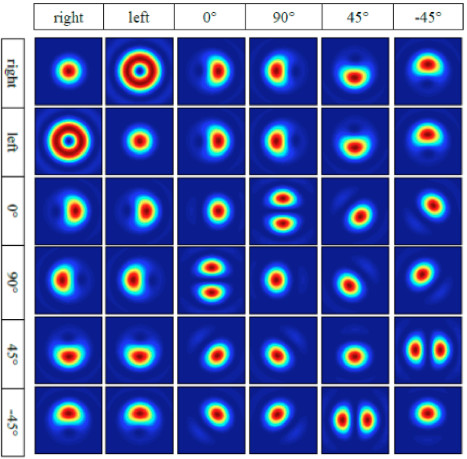
Figure 1
Family of spatial distributions, which can be generated by a biaxial crystal and polarizing optics. The upper horizontal axis indicates the input polarization state, and the vertical axis indicates the output polarization state.
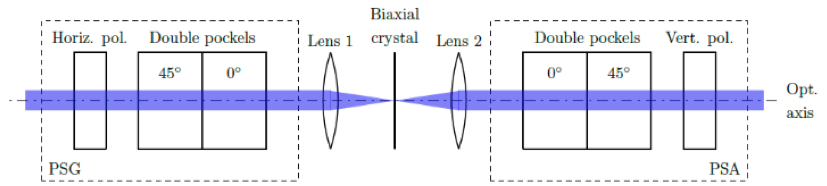
Figure 2
Beam shaper unit schematics, the biaxial crystal is placed in between polarizing optics to enable manipulation of both the input and output polarizations. In the optical path, prior to the crystal, a horizontal linear polarizer is used, after which a double Pockels cell acts as a polarization generator (PSG). This allows for the generation of any incident light polarization. Past the crystal, another double Pockels cell unit with a vertical linear polarizer acts as a polarization analyzer (PSA).

Figure 3
Typical light shapes used in the LiveCodim system.
Super-resolution using CODIM
The CODIM beam shaper is used as an add-on to a confocal module and the distributions are scanned on the sample, yielding several micro-images for each scan point. Due to the point scanning geometry, our system creates a reduced quantity of diffuse light, compared to the widefield-based technique.
The CODIM beam shaper generates compact, localized light distributions using the conical diffraction principle. Each micro-image contains a large number of high frequencies, close to Abbe´s limit (up to a factor close to 3 compared to an Airy pattern) (Figure 4).
These light distributions, projected on the sample, are analyzed using complex proprietary algorithms. This allows reconstruction of a super-resolved image, for general objects, with an improvement of resolution up to a factor of 2. Additionally, these algorithms, leveraging positivity constraint and sparsity, allow for the resolution to be improved even further, for adequate samples.
Ultimately, the conjunction of much lower distribution peak power, use of a high quantum yield camera, and a longer camera exposure time drastically reduce the peak power – and the energy – of light sent to the sample. Such is the reason for this method´s very low photobleaching and phototoxicity. This also avoids fluorophore saturation issues, making the method linear and quantitative.
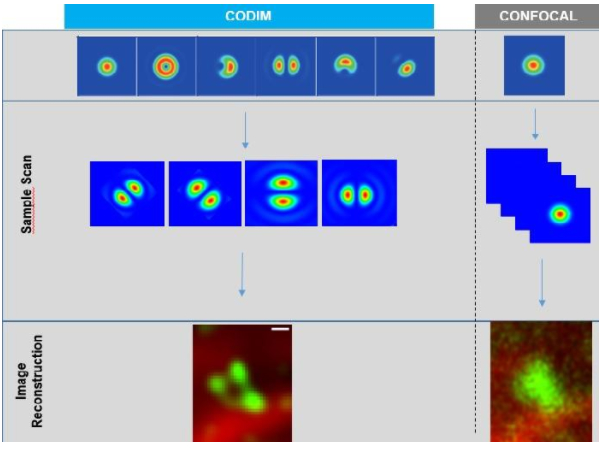
Figure 4
Measurement flow of LiveCodim system.
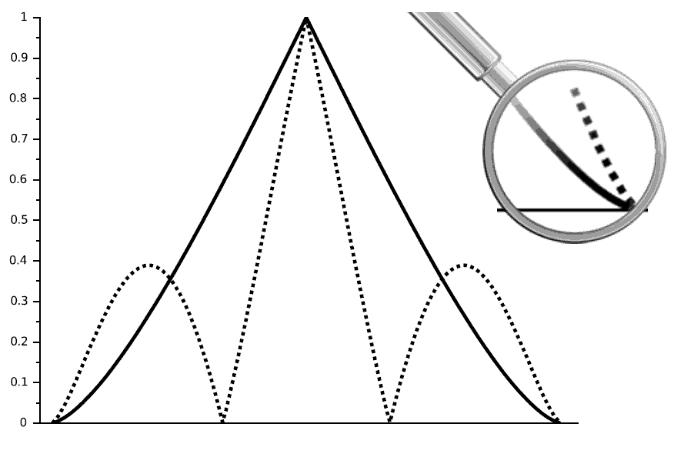
Figure 5
Mean Transfer Functions of LiveCodim (dotted line) distributions compared to Airy pattern (full line); close-up is a zoom on frequencies between 0.8 and 1 wavelength
Horizon 2020

BioAxial’s LiveCodim has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No. 811988 – LiveCodim.