Primary Assessment of Medicines with Quantitative Phase Imaging
Uncontrolled cell growth refers to cell multiplication without the usual regulation, which can lead to cancer. This rapid cell division can form tumors that may spread to other parts of the body, which is known as metastasis. Solid tumor metastasis is a major cause of cancer-related fatalities. To fight this, many cancer treatments focus on stopping the pathways that lead to uncontrolled cell growth and division.
Analysis of Cell Morphology, Migration, and Growth
Tackling cancer spread requires effective anti-metastatic medicines, called migrastatics. These compounds aim to stop tumor cells from moving to new areas. A quick test has been developed to find these potential migrastatics. Using a high-tech Q-Phase holographic microscope, scientists can closely examine cell morphology, migration, and growth across multiple fields. This analysis is crucial for understanding how cells interact with their environment and how changes in morphology can influence cellular functions. Changes in cell morphology can show behaviour changes, such as the transition from stationary to migratory states, which is important for cancer metastasis.
Developing PAMP
We have developed the Primary Assessment of Migrastatic Potential (PAMP) in vitro methodology to quickly evaluate potential migrastatics. This method looks at how these medicines affect cell movement in 2D cultures, which are simple and cost-effective setups widely used in research. The goal is to measure how well these medicines can reduce random cell migration while keeping the cells’ normal functions intact.
What We Found
Cell Line Selection: For our initial tests, we used well-known cell lines that are commonly used in cancer research. These cell lines help us consistently measure how potential migrastatics affect cell movement and other important behaviours.
Results with A549 Cells: We tested three medicines: Belumosudil (BEL), Midostaurin (MID), and Niclosamide (NICL). All three slowed down cell migration. BEL and NICL also changed the shape of the cells, making them rounder and less dense. While BEL and MID did not increase the number of invasive cells, NICL reduced these aggressive cells. MID shows promise for further studies, while BEL and NICL require more testing to find the best doses.
Results with HT1080 Cells: MID and NICL also slowed down cell movement in these cells. MID did not change cell morphology, but NICL made the cells denser and rounder. Neither medicine increased the number of invasive cells. MID is recommended for further testing, with NICL also suggested for additional trials at lower concentrations.
Methodology
The PAMP involves selecting the right tumor cell lines for detailed, time-lapse recordings using Quantitative Phase Imaging (QPI) from the Q-Phase microscope. This technology allows for precise, non-invasive imaging of cells and measures their dry mass, providing a thorough analysis of their migratory behaviour.
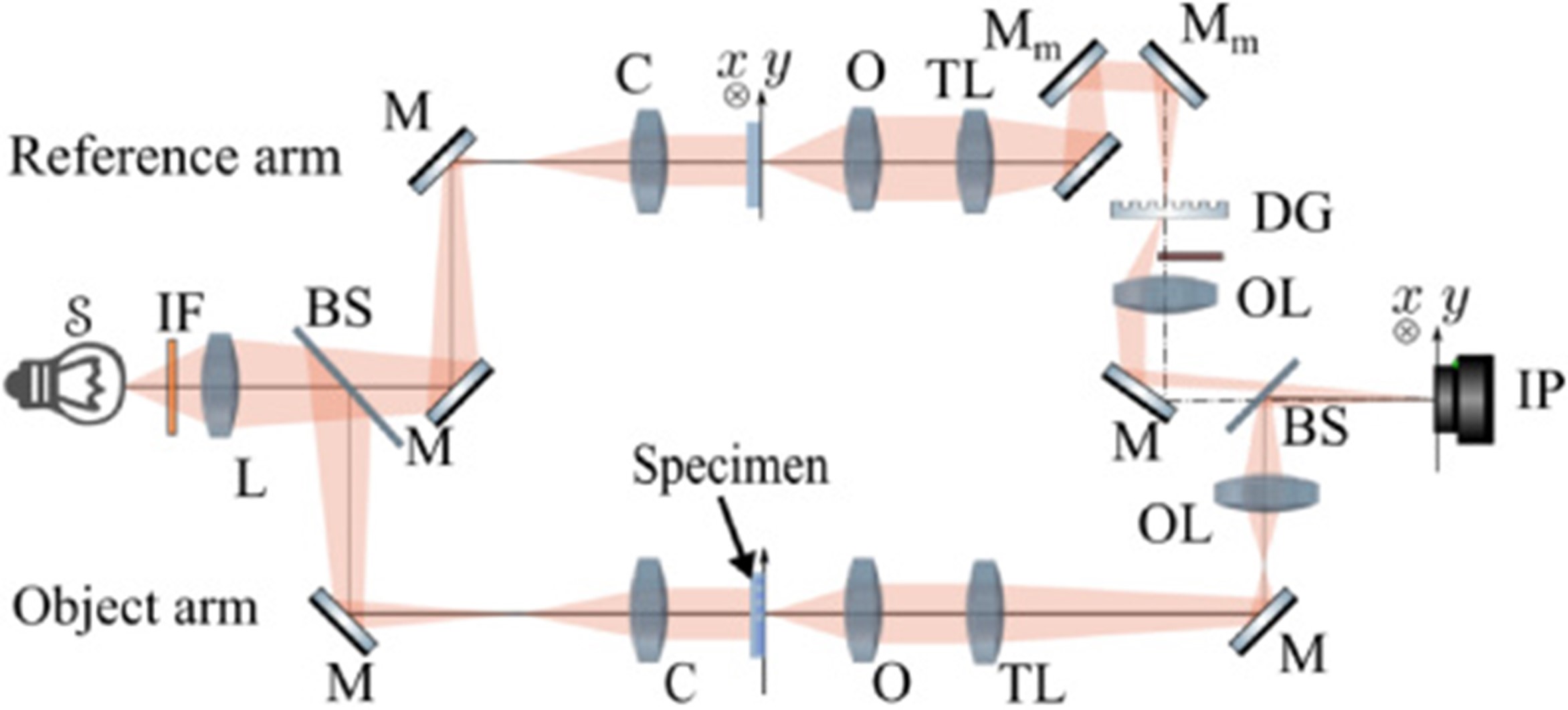
Optical setup of the coherence-controlled holographic microscope: S, light source; IF, interference filter; L, relay lens; BS, beam splitters; M, mirrors; Mm, movable mirrors; C, condensers; O, objective lenses; TL, tube lenses; DG, diffraction grating; OL, output lenses; IP, interference plane.
Conclusion
Based on the pilot study results, Midostaurin (MID) is a promising migrastatic agent for further research. The study also confirms that QPI is a reliable and cost-effective tool for examining the cell migration, helping us understand how potential migrastatics work.
Highlights:
- Development of PAMP Methodology: Researchers introduced the framework of Primary Assessment of Migrastatic Potential (PAMP) in vitro methodology, designed for rapid evaluation of potential migrastatics. PAMP measures the reduction in adherent cell random migration in 2D cultures while preserving undisturbed cell physiology, enabling efficient assessment of migratory behavior.
- Identification of Potential Migrastatic Candidates: The study identifies midostaurin (MID) as a promising migrastatic candidate for interfering with migration in both A549 and HT1080 cells. Additionally, belumosudil (BEL) and niclosamide (NICL) exhibit potential, with specific influences on cell speed, morphology, and invasiveness. Further testing with lower concentrations is recommended for BEL and NICL.
- Validation of QPI Technology: The research demonstrates the effectiveness of Quantitative Phase Imaging (QPI) obtained from the Q-Phase microscope. QPI enables precise, non-invasive imaging of cells, measuring their dry mass. The study suggests MID as a potential migrastatic for further preclinical validation, showcasing QPI as a reliable and economical technology for in vitro examination of cell migratory behavior induced by potential migrastatics.
The original article was written by Šuráňová, M. et al 2023.